These bonds are dependent upon the number of atoms sharing electrons and therefore are named using numeric prefixes to identify atom counts. The prefix mono- is generally omitted for the first element. Molecular Compounds also known as covalent compounds involve bonds between two non-metals. 
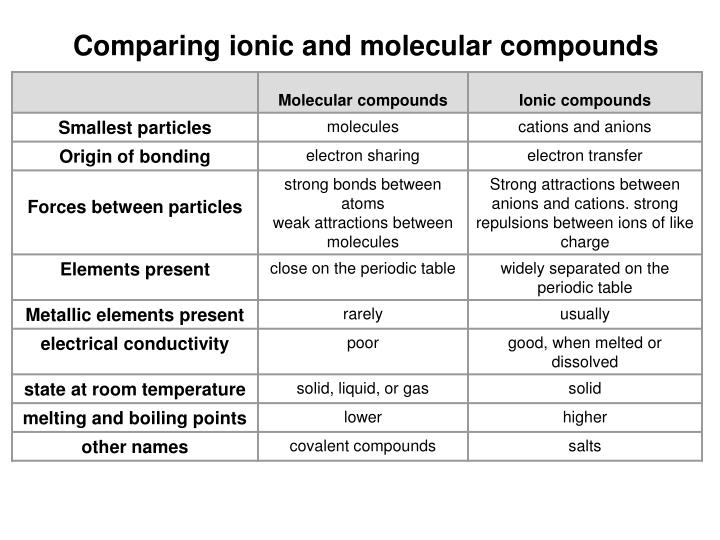
Compounds that do not contain ions, but instead consist of atoms bonded tightly together in molecules. An ionic compound is formed between a metal and a nonmetal (or polyatomic ions), and is held together through the attraction of opposite charges. Prefixes are used to indicate the number of atoms of a given element in a molecule ( mono- = 1, di- = 2, tri- = 3, tetra - = 4 …). Ionic compounds generally form from metals and nonmetals. Name of a binary molecular compound: name of the 1 st element of the formula + name of the 2 nd element with the suffix – ide. The word ion is omitted. If the metal is a variable charge metal, the charge is indicated in parentheses with Roman numeralsīinary molecular compounds are composed of 2 nonmetals Name of an ionic compound: name of the cation + name of the anion with the suffix – ide for non-polyatomic ions. 
They consist of 2 ions derived from 2 different elements: one metal element (a cation) and 1 nonmetal element or polyatomic ion (an anion) 12 explain why ionic and covalent network substances are solid (high melting points), and why covalent molecular substances can be solid, liquid or gas (low. A molecular equation is sometimes simply called a balanced equation. Most ionic compounds are binary compound. Definitions of molecular, complete ionic, and net ionic equations. Molecular Formula Empirical formula: the chemical formula with the simplest whole number ratio of atoms present in a compound Molecular formula. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
